For detection methods in CE, a distinction is made between on-column and off-column methods. In the on-column method, a part of the capillary itself is used as a measuring cell, while for the off-column method, an external detector is connected to the capillary output. However, this coupling is associated with dead volumes and dilution of the sample and thus usually with a corresponding peak broadening.
In principle, a wide variety of detection methods can be used, such as e.g. the optical methods UV / VIS and fluorescence detection and the electrochemical methods amperometric, potentiometric and conductivity detection. It is also possible to couple the CE with mass spectrometers.
UV-Detection
By far the most widely used detection method is UV detection. All commercially available CE devices are equipped as standard with a UV detector or a photodiode array detector.
The UV detection is carried out “on-column”, i.e. directly in the separation capillary, whereby the Lambert Beer law applies to the absorption of light:

The decisive factor for the detection strength of the detection is, according to Lambert Beer’s law, the small internal diameter of the CE capillary. The usual detection limits in CE with UV detection are in the lower ppm range (mg / l). An improvement of the detection strength can be achieved with special capillaries in which the inner diameter of the capillary is widened at the location of the detection window.
Indirect detection
The use of indirect UV detection technology significantly expands the range of application of CE, as it also allows UV-inactive analytes to be determined.
In the case of indirect UV detection, electrolyte systems are used which absorb UV radiation themselves at the selected detection wavelength and thus produce a constant background signal. The analyte ions displace electrolyte ions with the same charge sign (charge displacement) and thus cause a reduction in the intensity of the background signal.
The theoretical description of the change of the electrolyte concentration by the analyte ions results from the principle of electroneutrality and the Kohlrausch theory.
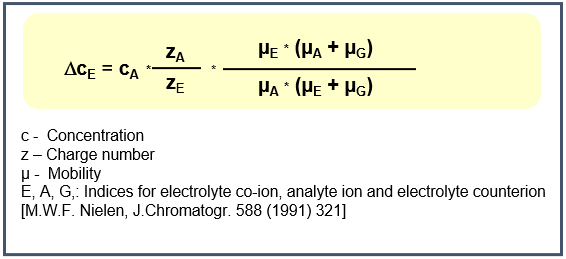
The concentration of the displaced electrolyte ions is directly proportional to the analyte concentration, making the indirect detection technique suitable for quantitative analytical determinations.
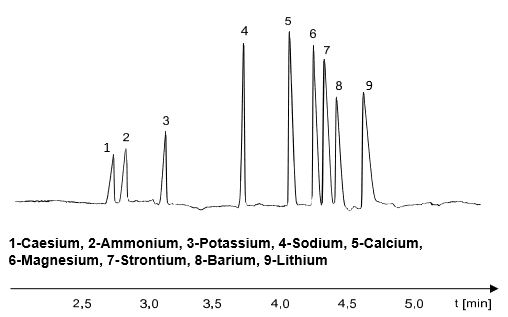
However, the choice of electrolyte concentration is limited by the strong UV absorption of the electrolyte ions upwards. While electrolyte concentrations of up to 100 mmol / l and more can be used for direct UV detection, indirect electrolyte detection limits the electrolyte concentration to approx. 10 mmol / l. As a result, the effects of electrodispersion increasingly occur, which often results in a triangular peak shape when there are different mobilities of electrolyte co-ions and analyte ions. This can be seen in the following electropherogram of the separation of UV-inactive cations with an imidazole electrolysis system. Imidazole has, under the conditions chosen here, an electrophoretic mobility that corresponds approximately to the mobility of sodium. The peak area is also proportional to the analyte concentration even in the triangular peak shape and can be used for quantification.